Now live! RTHM Direct – simplified medication access for Long COVID, ME/CFS and related conditions. Check it out
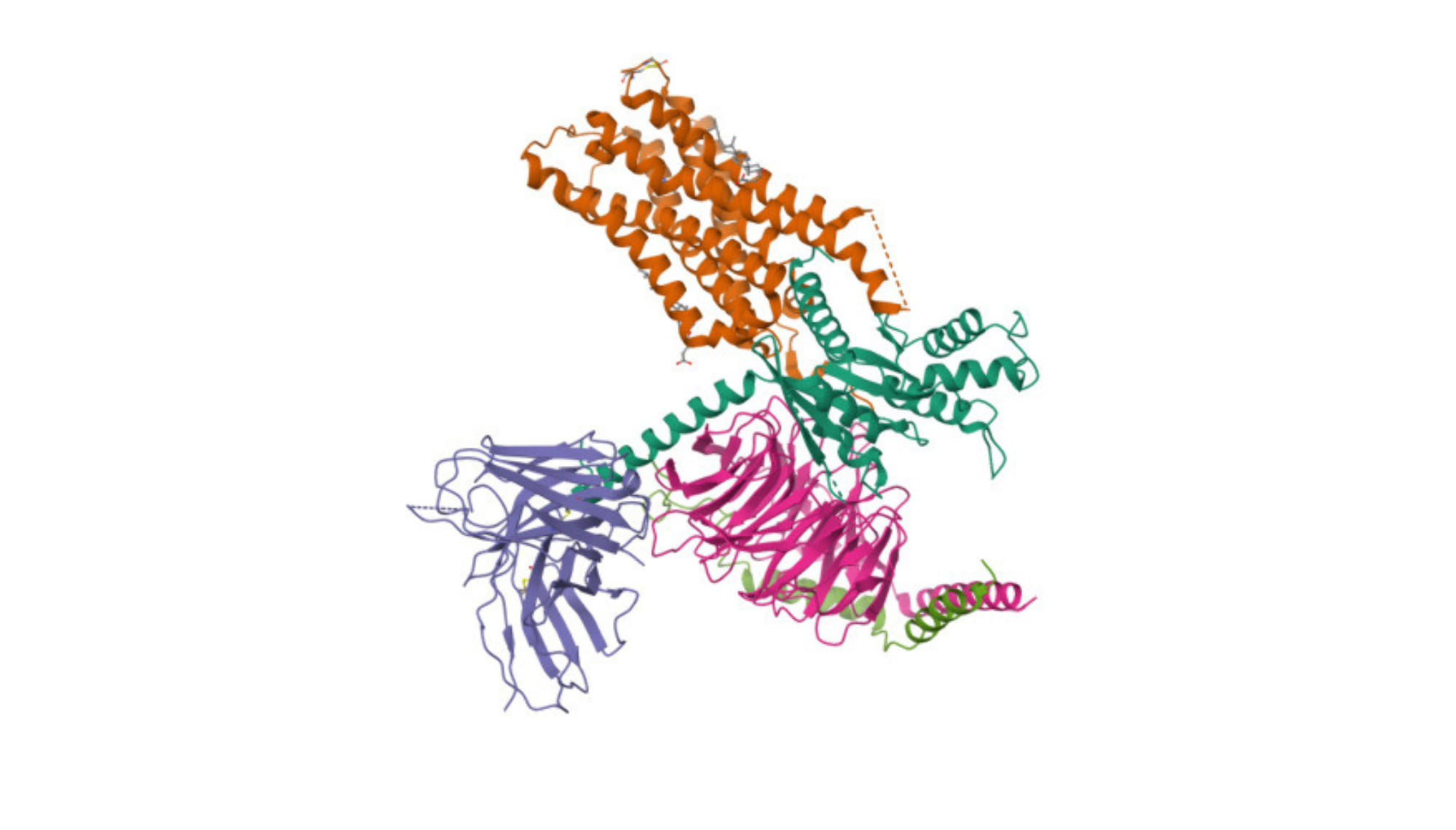
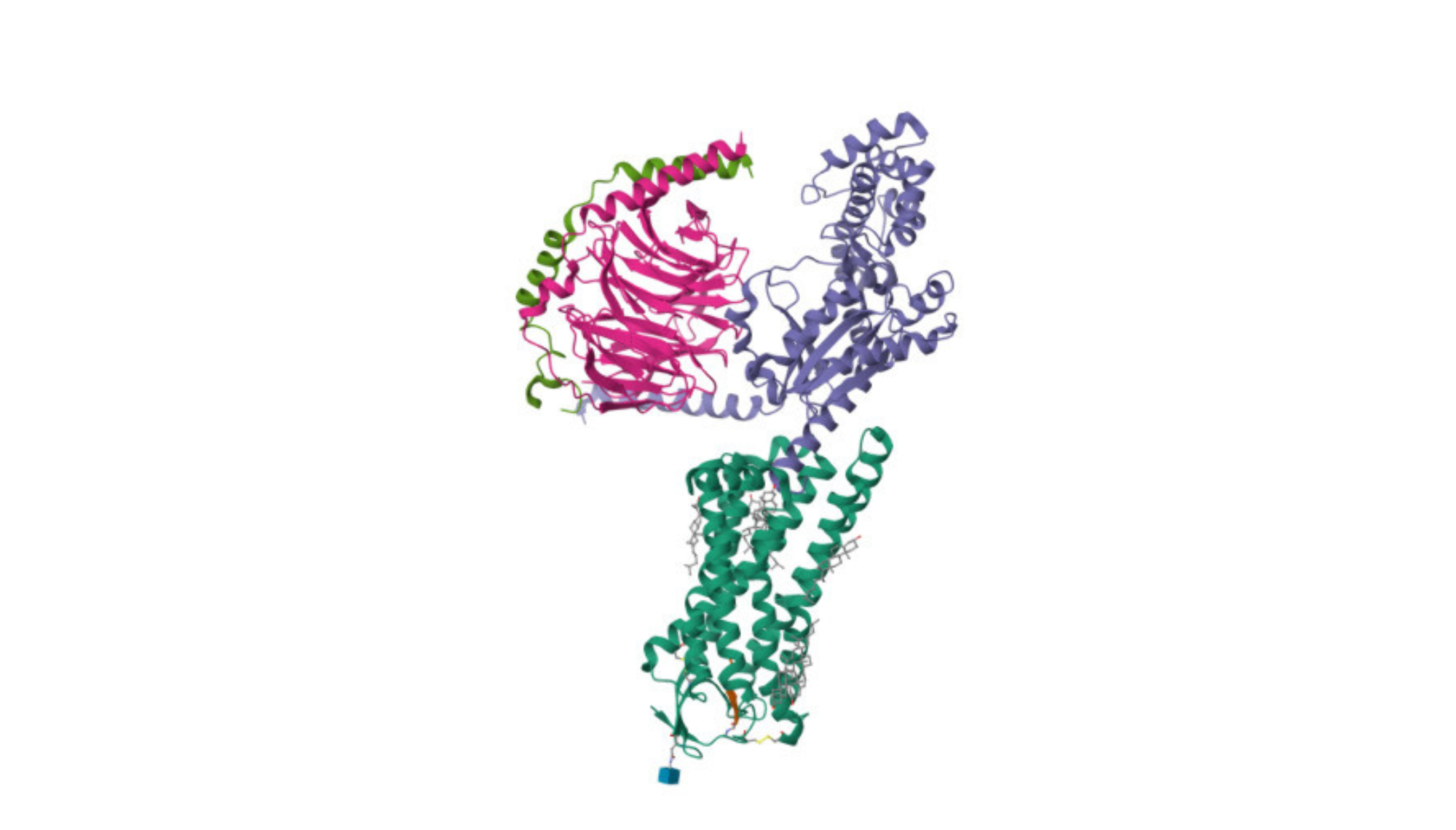
M1-R is predominantly expressed in the brain and plays a crucial role in modulating neurotransmission, particularly in cognitive processes, learning, and memory. Muscarinic cholinergic (M1) receptor is predominantly expressed in the brain and plays a crucial role in modulating neurotransmission, particularly in cognitive processes, learning, and memory. Its activation enhances neuronal excitability and is implicated in various central nervous system functions.
M1 Muscarinic Receptors (M1 mAChR) primarily couple to Gq/11 proteins, activating the phospholipase C (PLC) signaling pathway, leading to the formation of inositol triphosphate (IP3) and diacylglycerol (DAG). This results in an increase in intracellular calcium levels and activation of protein kinase C (PKC).
Functions of the Muscarinic Cholinergic M1 Receptor (M1-R)
The M1 muscarinic cholinergic receptor (M1 mAChR) is one of the five subtypes of muscarinic receptors (M1 through M5) that mediate various physiological responses to acetylcholine, a neurotransmitter. Here are the primary functions and characteristics of the M1 muscarinic receptor:
- G-Protein-Coupled Receptor (GPCR): The M1 receptor is a G-protein-coupled receptor, which means it activates intracellular signaling pathways in response to binding with its ligand, acetylcholine.
- Neurotransmission: M1 receptors are predominantly expressed in the brain and play crucial roles in neurotransmission. They are involved in mediating the excitatory responses to acetylcholine in the central nervous system (CNS).
- Cognitive Functions: The activation of M1 receptors has been associated with enhancing cognitive functions such as learning and memory. It’s thought that M1 receptor activation can help to improve cognitive performance, and as such, is a target for the treatment of cognitive impairments in disorders like Alzheimer’s disease.
- Smooth Muscle Contraction: Besides their role in the CNS, M1 receptors are also involved in smooth muscle contraction in various organs like the gastrointestinal tract, where they help promote gut motility.
- Secretion: M1 receptors are involved in stimulating glandular secretion. For example, they play a role in gastric acid secretion in the stomach, aiding in digestion.
- Cell Proliferation and Differentiation: Some research suggests that M1 receptors may also be involved in processes like cell proliferation and differentiation, which are fundamental for tissue repair and growth.
- Potential Therapeutic Target: Given its role in cognition and smooth muscle contraction, the M1 receptor is a potential therapeutic target for various disorders, including neurodegenerative diseases and gastrointestinal disorders.
- Signal Transduction: Upon activation, M1 receptors primarily couple to Gq/11 proteins, leading to the activation of phospholipase C, which in turn increases intracellular levels of inositol trisphosphate (IP3) and diacylglycerol (DAG). These molecules further activate other intracellular signaling pathways, leading to a variety of cellular responses including the release of intracellular calcium stores, which is fundamental for many of the processes mentioned above.
Autoantibodies to Muscarinic Cholinergic M1 Receptor (M1-R)
Autoantibodies to the M1 Muscarinic Cholinergic Receptor (M1 mAChR) have been associated with certain pathological conditions and may influence the receptor’s function, as evidenced by a few studies highlighted below.
Long COVID, Myalgic Encephalomyelitis/Chronic Fatigue Syndrome, and Dysautonomia
There is evidence of autoantibodies to the muscarinic cholinergic M1 receptor (M1-R) in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS). These autoantibodies may contribute to the dysautonomia symptoms experienced by individuals with ME/CFS, such as severe fatigue, dizziness, and orthostatic intolerance. Autoimmune autonomic nervous system imbalance, including the presence of autoantibodies against G protein-coupled receptors (GPCR), has been proposed as a novel mechanism in ME/CFS. (Malkova and Shoenfeld 2023) (Leobel et al. 2016) (Sunami et al. 2022)
Another study found that autoantibodies to M1 muscarinic cholinergic receptor 1 (CHRM1) were detected in a significant number of patients with CFS. The presence of these autoantibodies was related to CFS symptoms, suggesting that subgroups of CFS are associated with autoimmune abnormalities of CHRM1. Notably, patients with positive autoantibodies to CHRM1 had higher mean scores of ‘feeling of muscle weakness,’ among other symptoms like ‘painful node,’ ‘forgetfulness,’ and ‘difficulty thinking,’ although the latter symptoms did not reach statistical significance. (Tanaka et al 2003)
Autoimmune Conditions
One study investigated the activation of nitric oxide signaling in submandibular glands by antibodies from patients with primary Sjögren Syndrome, which is an autoimmune disease. The study demonstrated that these antibodies can activate nitric oxide signaling through the interaction with muscarinic acetylcholine receptors, including M1 receptors, in the submandibular glands. This suggests that autoantibodies to M1 receptors may have a role in the pathogenesis of certain autoimmune conditions. Further research is needed to fully understand the implications of autoantibodies to M1-R. (Leiros et al. 1999)
Modulation of Cholinergic Neuromuscular Transmission and Myasthenic Disorders
Autoantibodies against the M1 mAChR can potentially affect synaptic compensatory mechanisms. For instance, the M1 mAChR modulates cholinergic neuromuscular transmission by linking to P/Q-type voltage-gated calcium channels (VGCC), which may partially compensate for reduced calcium entry.
In Lambert-Eaton Myasthenic Syndrome (LEMS), a disorder often associated with small-cell lung carcinoma (SCLC) and characterized by a deficit in acetylcholine release from motor nerve terminals, autoantibodies against the M1 mAChR were detected in a portion of patients. These autoantibodies were found in both anti-VGCC-positive and anti-VGCC-negative LEMS patients, highlighting their potential to affect synaptic compensatory mechanisms, more so in LEMS than in Myasthenia Gravis (MG). (Takamori et al. 2007)
These findings suggest that autoantibodies to the M1 Muscarinic Cholinergic Receptor could play a role in the pathology of certain disorders, potentially impacting synaptic transmission and contributing to the symptomatic manifestation of these conditions.
Learn more about autoantibody testing at “RTHM Lab Now Offering GPCR+ Autoantibody Panel” and visit our Lab Page to order your GPCR+ Antibody Panel today.
Featured image courtesy of RSCB Protein Data Bank

Get updates
Join our mailing list